index


This page, as with others, is for both personal reference, as well as general interest to others.
Years ago in 1992 I used my Web pages to look up company data: Meter readings and values, and inventories,
including company equipment. I looked up this information in any town by plugging in a big heavy company laptop.
I was in the process of being sued when I and my attorney decided to withdraw company information.
But I consider my efforts at the time, to have invented the "Cloud".
In 1992 I was one of the first to have invented a Cloud of sorts.
Now, I still keep records, and this page is one of them.
I keep information about my RV batteries that I have had, or that I still have, or that I will have.
Now, I do not need to be in a town, nor to have a laptop;
I just simply tap on my cell phone and see exactly what I have, any time or any place.
The concept is the same as many years ago; just easier now.
And no one can sue me as this is personal information.
 I took the two batteries out of the RV. With wires, I connected a large 25 watt resistor
across the battery posts.
The resistor is about 2 ohms, and pulls exactly 5.26 amps initially.
The resistor was placed in a gallon bucket of water.
The resistor was kept submerged at all times.
The horizontal axis is hours.
I took the two batteries out of the RV. With wires, I connected a large 25 watt resistor
across the battery posts.
The resistor is about 2 ohms, and pulls exactly 5.26 amps initially.
The resistor was placed in a gallon bucket of water.
The resistor was kept submerged at all times.
The horizontal axis is hours.
Battery-1
Left
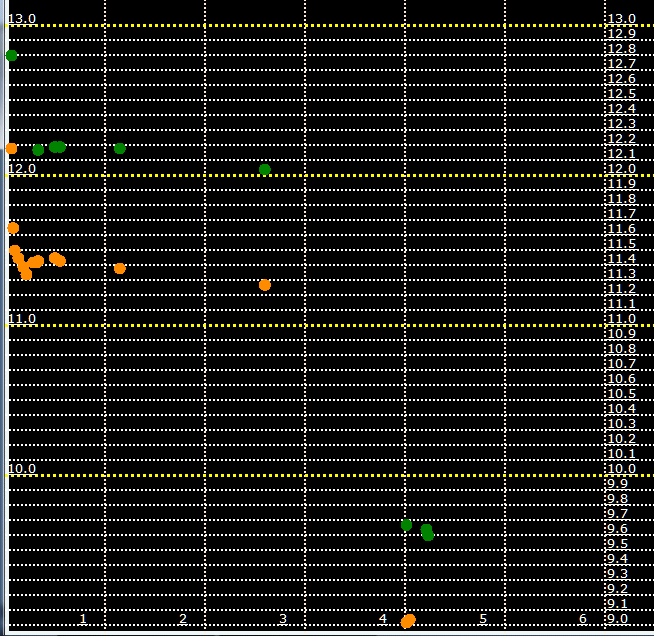
The yellow points are under load, voltage measured at the posts with the resistor connected.
The green points are with the resistor temporarily lifted, and with the battery recovering.
The battery resistance can be estimated by the difference of these two readings: the yellow points minus the green points.
And the amperage is not constant but decays down from the initial 5.26 amps.
I took a static reading by temporarily increasing the amps to 13.48A
and obtained a battery resistance of 0.058 ohms.
There are two types of battery posts on this battery:
One is a big Lead Post, useful in large current demands such as boat trolling or starting.
I can not imagine why a deep cycle battery would even have one of these.
The other is a stainless steel stud. It is good for small electrical utilities.
There is a 0.66 volt difference between the two when using 5 amps!
Fixed the problem by bridging the Lead post to the Stud Post with a flexible "bar".
I bridged the negative two posts together, and also the two positive posts together.
Evidently, stainless steel does not bond well with lead in the construction of the battery.
I was in disbelief, but the phenomenon is really for real, as I went over and over the situation many times.
Now I know why these batteries come with a big lead post:
It is to repair the nearby stainless steel post.
This battery is over 5 years old; new in 2009.
Although my bucket of water was hot, in less than 4 hours this battery was totally dead.
Having a hot bucket of water is no conciliation for pitiful operation.
As you can see, the aged battery now shows less than 20 Amp-Hours.
And I was not around to observe, or get readings, near the "knee" when the battery took
a fast nose dive. It took a nose dive at about 3.5 hours. But it doesn't matter.
This battery is just pitiful.
That explains why the TV would last for only about 3 hours with this battery.
It also explains why the solar panels could recharge this battery back up in only a couple of hours.
The battery would charge back up to 13.48 volts in no time at all. Something was wrong.
This battery is just plain pitiful. This battery is history.
Battery-2
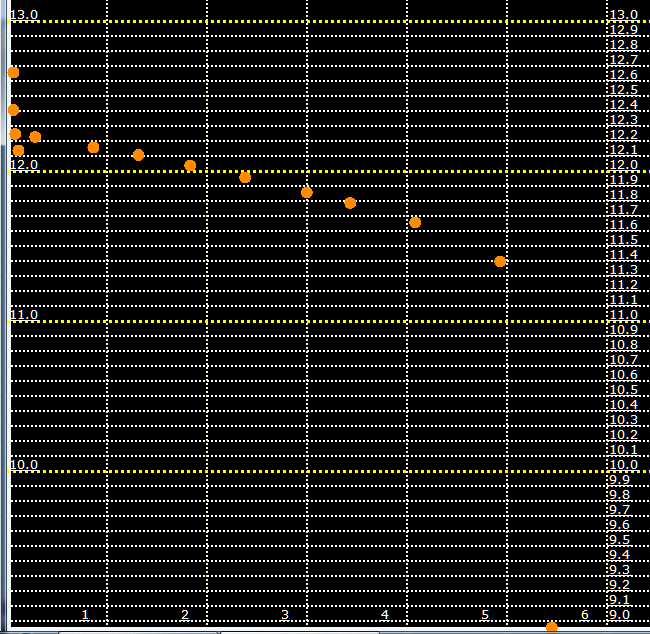 Voltage at the posts under an initial 5.0 amp load.
Voltage at the posts under an initial 5.0 amp load.
Initial battery resistance is 0.034 ohms.
34 mOhms internal long-term Resistance is kinda OK.
At least the physical posts were constructed OK on this battery,
and there was no resistance under the small Stud-Post, what so ever.
The battery ran for 5 hours before "crashing".
The battery measures less than 25 amp-hours.
This battery was originally a 85 amp hour battery three years ago.
Not too good...
 Sitting here looking at the bucket of water, I see a another way to obtain the amp-hours.
Sitting here looking at the bucket of water, I see a another way to obtain the amp-hours.
And not by using the water to just cool the resistor...
Amp-hours is a measure of the quantity of electric charges, or coulombs.
It is a convenient term when measuring battery reserves, capacity, or specifically coulombs that are under the action of a certain voltage.
Watt-Hours is perhaps a more useful term! It is Energy. A main building block of science.
The two are related: Amp-Hours times the voltage is Watthours.
In the case of these 12 volt batteries, Watthours are approximately 12 times the Amp-hours.
For example, a 100Ah battery is also known as a 1200Wh battery (roughly).
Anyway, the energy given to the bucket of water is easy to determine.
We must know the quantity of water, and the temperature difference.
The container of water must be well insulated.
By measuring the old and new water temperature, one would have the total energy.
The energy is known from the thermal capacity of water.
The I^2R Energy is the same as the water heat energy.
Now, one does not need the clock, nor to have a steady current.
W=(P)dt=(i^2 * R)dt, I=q/dt
I=(W/RT)^.5
IT=AmpHours=T(W/RT)^.5 =(TW/R)^.5
I lied; Still need the clock for time T.
But Time is well established, Energy W is accurately known, Resistance is accurately known.
Cool!
The State Of Charge of Lithium Ion is difficult to directly observe.
Lead Acid batteries indicate their state of charge by their output voltage.
For example, fully charged may be at 12.60 volts, half at 12.06 volts, and zero at 10.50 volts.
A Lithium Ion, on the other hand, maintains its output voltage constant, until a "crash",
with a very sharp knee.
Of course, a good Controller can "coulomb count" always keeping track of input Amp-Hours and output Amp-Hours, but it is a guess.
So, Lithium can only be measured from a complete course of 100% to 0%, and no partials in between.
A bucket of water may be useful for measuring Amp Hours because you do not need a clock.
But you do need to go to completion: 100% to 0%.
New AGM Battery

 This battery failed in less than 5 months. Capacity continually dropped until now only about 10AHr.
This battery failed in less than 5 months. Capacity continually dropped until now only about 10AHr.
Cost to much to send it back.
The battery that it was replacing lasted 6 years.
This battery was treated well: Floated at a 13.2v to 13.6v. But discharged to 11.7 a couple of times.
Under a homemade test, a fully charged battery drops from 13.00 volts
to 11.78 volts under an 11 amp load.
This AGM battery is crap! No more AGM batteries for me even if they are not made in Asia.
Have personally seen no evidence that AGM batteries are as good as Wet Cell.
Cheaper RV batteries are cost effective.
 Purchased this battery locally, at Ron's RV.
Purchased this battery locally, at Ron's RV.
Initial voltage at terminals was 12.3 indicating the battery about 75%.
If the store owner has not left it in this condition too long, it should have no sulfate.
 The date code shows that the battery is mine: My initials are "CAP", Cecil Alan Pennock.
Just kidding; Wierd things happen all the time.
For example, before custom car plates in the 50's,
my Dad had a licence plate - quite by accident - that was "Cec". Coincidently, he went by that name!
But I do not go by "CAP".
The date code shows that the battery is mine: My initials are "CAP", Cecil Alan Pennock.
Just kidding; Wierd things happen all the time.
For example, before custom car plates in the 50's,
my Dad had a licence plate - quite by accident - that was "Cec". Coincidently, he went by that name!
But I do not go by "CAP".
Anyway, The Months indication is the first letter.
"A" is for Jan, "B" is for Feb, "C" is for Mar, etc.
"F" is for the 6th month or June. The next letter "5" is for the year, or 2015.
The factory delivered this battery 2015.June. Bought on 2015.09.20. Three months old.
The next three letters signify who is going to own it.
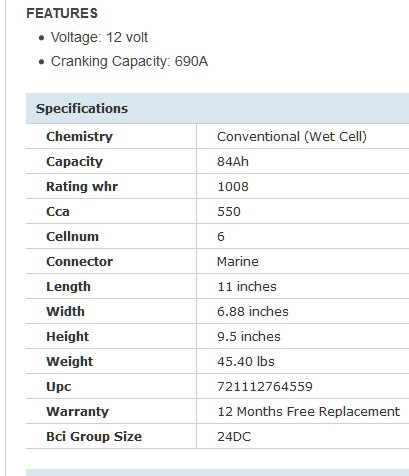 84 AHr
84 AHr
Actually, it is not 84. The listed 1008wHr suggests 80AHr.
This is the only battery that I plan to keep. And it is because it is a Wet-Cell.
I actually want a battery that you can see inside.
I want to be responsible for the water level. I do not trust AGMs or Gels.
I want to actually see the little bubbles from gassing, if any.
I feel that after 5 or 6 years, sealed Lead Acids will need water.
Unlike those things, this Interstate will be "serviceable".
I am not ready to go to Lithium yet.
 Temperature
Temperature
Temperature seems to be the enemy of any type of battery.
But for Lithium Phosphates the high temperature is deadly, and ridiculously stressful.
For example, the Aging Curve for AmpHour capacity will be accelerated
to the tune of doubling for every 20F degrees increase from the test conditions of 77F degrees.
So if the operating temperature is 97F degrees for one year,
the expected Aging will be permanently doubled, and the AmpHours are cut in half.
That is huge! And the derating curve is permanent!
So Lithium Phosphate can not be stored outside, or in the sun, or on the tongue of a trailer.
Neither can they be used in my motorcycle, or in most motorcycles, with high temperatures under the seat.
In real world cases Lithiums work fine for the first year, but in all cases of heat,
begin to see drastic reductions of AmpHours.
 EMP
EMP
I can find no measurements of damage from a nuclear air burst.
But I know that Lead Acids are very tolerant to EMP,
and these old style batteries will still be around.
Batteries are an extremely important component to RVs, as well as cars and trucks.
There is no evidence that Lithium can withstand an EMP attack. They are very sensitive to voltage.
Again; Lithium must be kept inside a hardened inclosure, such as with a metal skinned RV.
I do not have one, but Lithiums in Airstreams may still work.
 Cost
Cost
So far, it is more immediatly practical, and probably cheaper,
to go with a $120 dollar 100AH wet Lead Acid battery than
a $1200 Lithium. The pros and cons go on forever, and are highly tailored to specific needs.
 No dought eventually, I will be going Lithium. Looking forward to it.
But first I must see how my new Interstate holds up.
...And also how the several different Lithium chemistries pan out.
No dought eventually, I will be going Lithium. Looking forward to it.
But first I must see how my new Interstate holds up.
...And also how the several different Lithium chemistries pan out.
 Interstate battery:
Interstate battery:
Here are some readings with a new meter: Model AE300.
It can not directly measure AmpHours.
No one, to my knowledge, has invented a meter yet that can measure AmpHours.



I put in the wrong CCA. It is actually listed as 550A CCA,
not the 690A Cranking Capacity that I mistakingly told the meter.
To make matters worse, the meter is reading Cranking Capacity (690A) and not CCA.
What a wierd coincidence: the meter reads exactly what I typed in.
Despite that problem, this new meter says battery is more new than new. Not possible!
The volt meter reads 5 mV low, which is OK.
2015.09.30 to 2017.09.19 is two years old. ...And shows no degradation?
I also disconnected the battery, and took isolated readings. No change.
 The Everlast
The Everlast
Battery-Right, Battery #2
2012.Feb to 2017.09 is 5.7 years.



Shows double the internal resistance of the first battery: 7.95mOhms, 0.0079 Ohms.
This battery needs to be replaced.
RV Battery A, #1, Left
Interstate study:
Battery-Left, Battery #1.
 Got the bucket of water out. Found two one ohm resistors.
Wired them in series, and added a little incandescent bulb in parallel, for esthetics.
The glow on the left is the incandescent under the water.
Got the bucket of water out. Found two one ohm resistors.
Wired them in series, and added a little incandescent bulb in parallel, for esthetics.
The glow on the left is the incandescent under the water.
 Placed everything in the bucket of water. Everything reads about 1.5 ohms.
Aiming for about 6 amps.
The standard 20C would be in this case 4 amps, but I want to hurry this up.
Placed everything in the bucket of water. Everything reads about 1.5 ohms.
Aiming for about 6 amps.
The standard 20C would be in this case 4 amps, but I want to hurry this up.


Waited for exactly 13:00 on my watch to run the test.
The resistors and light bulb pull 5.89 Amps,
and the voltage suddenly dropped from 12.87v to 12.24 with this load.
This represents a huge internal DC resistance: 0.63 volt / 5.89 amps is 0.107 ohms.
That is 107 mOhms! Measured again with the same outcome.
Nowhere near the internal dynamic resistance of 4.16 mOhms as read by the new meter.
The voltage should not have dropped. It should have stayed above 12.6v for a while.
But, for whatever it is worth, the voltage was solid at 12.1 volts for several hours.
It is like there is a top surface charge above 12.1v, and it is really soft.
I thought surface charge was only above 12.6v, so that makes me have a bad learning curve.
At 20:15 the voltage was 11.36 volts and getting close to collapse.
It had been 7.25 hours sense the start at 13:00.
7.75mOhms and 236 CCA, DC internal resistance was .3volts/5.6Amps = 50mOhms
The bucket of water was very warm.
At 22:35 the voltage had collapsed to 6 volts.
Next time, I need to put a monitor clock on it so that I do not miss that critical knee.
It had been 9.5 hours, and reads 10.66mOhms, 3.60 CCA.
(Full capacity reads 690CCA on the meter, Rating is 550CCA and 690A.)
The Amps did not stay at 5.89Amps the whole time,
and I am going to estimate a more realistic average:
I will call it 5.63 Amps.
And I know that the collapse was between 20:15 and 22:35.
So the AmpHours are between 53.5AH and 40.8AH.
This Interstate battery is rated 80AH, and fails my test, at least at this discharge rate.
Useful?
At 20:15 the CCA(236) was 0.4 of Full CCA(590). 0.6 was 7.2Hrs
Implying the whole time should have been closer to 12 hours instead of the 9.5 hours at most.
Therefor the CCA is, so far, not useful for anything, at least not to do with measuring AH.
So far, there is no known way to measure AH without getting out the bucket of water and Coulomb Counting.
The next morning the battery had recovered on its own to 11.80 volts.
With a reading of 7.93mOhms, 268CCA.
During the night on it own the ohms went from 10.66mOhms to 7.93mOhms, and the CCA from 3.6 to 268.
I began another run to get the last remaining Current out.
I pulled about 5.38Amps of additional current for 20 Minutes.
Now the battery is really dead. Bone dry.
So, with this little bit of a rally, I would estimated the AH at the upper figure of 53.5AH.
 Now I will measure the CCA going the other way: during a CHARGE.
Now I will measure the CCA going the other way: during a CHARGE.
Battery measured out of circuit, with 30 seconds of rest.
Putting in 12.0 Amps, and after a minute the meter reads 8.72volts, 14.70mOhms, 30.72CCA.
 After half an hour, still 12 Amps charging. Meter reads 12.26volts, 7.84mOhms, 312CCA.
After half an hour, still 12 Amps charging. Meter reads 12.26volts, 7.84mOhms, 312CCA.
This is about 6 AH input.
 After one hour, 12 Amps charging. Meter reads 6.42mOhms, 12.46volts, 403.2CCA.
After one hour, 12 Amps charging. Meter reads 6.42mOhms, 12.46volts, 403.2CCA.
This is about 12 AH input.
With mathematics you can map anything to anything.
Already, it is plain to see, there is no linear relationship between CCA and AH.
Looks like there is a step, or floor, of about 300CCA before AH can start to be measured.
So far, a formalism would dictated 690CCA represents 53.5AH at the top.
And at the bottom, either an aggressive exponential, or a DC offset step at the first.
 After two hours, 11 Amps charging, 12.71v, 7.46mOhms, 371.3CCA.
After two hours, 11 Amps charging, 12.71v, 7.46mOhms, 371.3CCA.
After a minute of rest: 12.60volts, 7.44mOhms, 361.9CCA.
...So from now on, a one minute of rest.
This is about 22 AH input.
An analyses shows the CCA going back down from an hour ago.
I have no idea what this means, other than I should dump lead acid batteries!
They are too spooky at a distance.
 After 2.5 hours, 11Amps, 12.69volts, 7.28mOhms, 378.5CCA.
After 2.5 hours, 11Amps, 12.69volts, 7.28mOhms, 378.5CCA.
This is about 28 AH input.
 After 3.0 hours, 9A , 12.84V, 7.19mOhms, 398.6CCA.
After 3.0 hours, 9A , 12.84V, 7.19mOhms, 398.6CCA.
This is about 32 AH input.
 After 6.0 hours, 6A, 13.34v, 4.35mOhms, 749.1CCA
After 6.0 hours, 6A, 13.34v, 4.35mOhms, 749.1CCA
This is about 53 AH input.
53 AH is about the same output electricity (coulombs) that came out on the Discharge Run.
But the 4.35mOhms Charging Run can go lower to 4.16mOhms Discharge Run.
This suggests we need to do more charging.
The CCA(749.1CCA) Charging Run is already above the Discharge Run(690.0CCA)
This suggests the battery is charged.
Clearly, the battery is not charged due to the 6A charging current.
There is some bubbling in number 5 cell. The rest are not quite charged.
 After 6.5 hours, 4.9A, 13.78v, 4.78mOhms, 756.8CCA
After 6.5 hours, 4.9A, 13.78v, 4.78mOhms, 756.8CCA
This is about 55 AH. Disconnecting charger...
 One hour of disconnect. 13.21v, 4.65mOhms, 678.4 CCA.
Reconnected.
One hour of disconnect. 13.21v, 4.65mOhms, 678.4 CCA.
Reconnected.
 After 2 final hours of charging, (3A, 15.0v charger), 13.63volts, 4.99mOhms, 701.0CCA
After 2 final hours of charging, (3A, 15.0v charger), 13.63volts, 4.99mOhms, 701.0CCA
All cells are bubbling. Batt is charged, perhaps a little over.
This is about 61 AH.
Disconnected Charger.
Volts down, ohms up, and CCA down, are all getting "worse".
With only 3A current, parameters must be returning to Rest or "no charge" condition.
Therefor all readings are misleading if a lot of input current has been recently active.
Probably due to large surface effects.
 After 3 hours of rest: 13.19v, 4.64mOhms, 676.8CCA
After 3 hours of rest: 13.19v, 4.64mOhms, 676.8CCA
Surface charges are dissipating...
After 12 hours of rest: 13.06v 4.62mOhms, 659.6CCA.
The biggest Rest-Changer is CCA: 2.6% change in CCA
The question remains: How is this related to AH?
 Internal DC Resistance.
Internal DC Resistance.
I connected the two 1 ohm resistors across the battery. They pull and measure 5.80 amps.
The voltage dropped from 13.06 to 12.82, a difference of 0.24 volts.
13.06-12.82/5.80A = 41.4 mOhms.
This is a huge reduction from the original state of 0.107 ohms or 107 mOhms.
There was a benefit from something that is never to be done: discharging a Lead Acid to zero.
That is less than half of what it was!
I am wondering what would happen if I took a crescent wrench across the terminals for a second.
Besides hundreds of amps and a big arc, I might inhance this process and dislodge some sulfate from the plates.
OK, but I do not want a bite taken out of my crescent wrench. It looks unprofessional.
At work, I had plenty of wrenches, screwdrivers, and pliers with gouges taken out;
It was embarrassing as people came into the shop...
Instead, a foot of 14 gauge wire should do, and I will get my sunglasses.
12.94v-12.74/5.80A = 34.5mOhms
12.89v-12.70v/5.8A = 32.8mOhms
It is getting better! It works.
RV batteries are never subjected to this heavy draw effect.
Car batteries are! And all the time!
Every day, for a half second, a car battery starts a car with hundreds of amps.
From what I have found, a 1/16 of a second of 600 amps would be beneficial.
This is only 0.01 AH, and that is nothing.
Something like this would be hard to technically implement.
I already have Equalization implemented in my controller, and it is was easy,
and is implemented everyday for about a minute.
But not desulfination, if that is what this is.
None of this has to be worried about with Lithium Phosphate; ...Just money.
Lithium is more like a glass piggy bank: You can see what goes in and what goes out.
RV Battery B, #2
 On 2017.09.25
On 2017.09.25
Bought this Interstate to replace the 25AH EverLast.
This new Interstate is going into the Right Position, or Battery-Right, or Battery #2
Now, I have two identical Interstates. One at 53AH, from the study above, and this new one at 80AH.
... or at least it should be.
 Batt #2
Batt #2
Here is the Date Code: I78FP
The "I" indicates the 9th month.
The "7" indicates 2017.
So, it was just shipped from the factory this very month (September).
 Standard Group 24
Standard Group 24
690A MaxCCA
Reserve Capacity: 140
The battery will supply 25 amps for 140 minutes, which is 58.3AH at this high rate (25A).
The 58.3AH is compatible with the 80AH;
but at the lesser rate, with the standard .05C (or 20 hours), of 4 amps.
There are two standards that measure the same thing sort of, but from different perspectives:
(Both involve "Coulomb Counting" of Amps times Time. But, when testing, either variable can be held constant.)
 Reserve Capacity AH.
Reserve Capacity AH.
Reserve Capacity never changes the amps (25 amps), but the time changes.
 Amp Hours, AH.
Amp Hours, AH.
Amp Hours never changes the time (20 hours), but the amps change.

 State of Charge: 12.66volts
State of Charge: 12.66volts
Internal Dynamic Resistance: 4.42mOhms
I typed in the wrong CCA. It is rated 560CCA and not 550CCA as was the old Interstate.

 Here is another set of readings, after setting half hour.
Here is another set of readings, after setting half hour.
State of Charge: 12.67volts
Internal Dynamic Resistance: 4.41mOhms
Which is about the same as the other Interstate (53AH)
CCA measures 621.3CCA. Better than advertised, 560CCA advertised.
But CCA does not have any relevancy for an RV application.
Nor is there any correlation with AH, as far as I can determine.
Internal DC Resistance



Internal DC Resistance:
Unloaded voltage is 12.68 volts,
nearly the same as other meter (batt condition meter state of charge 12.67v)
The load is two one ohm resistors in series:
They pull 5.65 amps. They were only engaged for a second.
And the loaded voltage is 12.54 volts.
Therefore the internal DC Resistance is: 12.67v - 12.54v/5.65A = 23.0mOhms. Great!
That is better than the old Interstate at 32.8mOhms.
Batt2, BattR (the new battery)
5.65Amps is the Load, PreVolt and After volts are averaged.
13.01v-Fluke 12.90v-Analyser 4.73mOhms 616.4CCA
PreVolt 5.65 Amps AfterVolt DC Resistance
12.90v-F 12.71v-Load 12.84v-F 28.3mOhms
12.84v-F 12.67v-Load 12.82v-F 28.3mOhms
12.82v-F 12.67v-Load 12.80v-F 24.8mOhms
12.80v-F 12.66v-Load 12.79v-F 23.9mOhms
12.78v-Analyser, 4.72mOhms, 599.2CCA
Batt1, BattL
5.65Amps is the Load, PreVolt and After volts are averaged.
13.01v-Fluke 13.00v-Analyser 4.27mOhms 700.3CCA
PreVolt 5.65 Amps AfterVolt DC Resistance
13.00v-F 12.79v-Load 12.98v-F 35.4mOhms
12.98v-F 12.82v-Load 12.96v-F 26.5mOhms
12.96v-F 12.81v-Load 12.95v-F 25.7mOhms
12.95v-F 12.79v-Load 12.93v-F 26.5mOhms
12.95v-Analyser, 4.28mOhms, 691.0CCA
According to the store bought analyser meter, the Old battery is better than the new.
Better CCA (599.2 vs 691.0), Less change in CCA (17 vs 9).
Better Meters Dynamic Resistance (4.72 vs 4.28).
If that absurd set of readings is correct then my Shorting Shock Treatment really works.
The only readings that makes sense are my own readings of the DC Resistance:
They are ever so slightly worse in the old battery, like they should be...
RV Battery B, #2, Right
Battery B2, Battery Right, or Battery #2
Also, this is the "new" battery, placed here in the #2 position a couple of years ago.
Actually, its date is 2017.09 September.
After staying in a Walmart, decided to take a look at the #2 Lead Acid battery.
It can stay up for only a couple of minutes before dropping to below 12 volts.
It either has a bad cell, or has ridiculously low AmpHours.
 After charging for a day at 13.40 volts, the battery was disconnected.
And almost immediatly it dropped to 11.23 volts. It has to have a bad cell...
And, it has only 16 CCA instead of 600CCA. Something is bad wrong!
After charging for a day at 13.40 volts, the battery was disconnected.
And almost immediatly it dropped to 11.23 volts. It has to have a bad cell...
And, it has only 16 CCA instead of 600CCA. Something is bad wrong!
 Rechecked the meter. It is even worse: 11CCA.
Rechecked the meter. It is even worse: 11CCA.
And that resistance is super bad:123 mOhms, should be about 4 or 5 mOhms.
Rechecked again; 8.6 CCA. Each time getting worse.
 So, I grabbed a couple of steel tools: a crescent and a socket bar. I shorted the posts together.
I was amazed to see just a tiny spark. I did not even need to blink, and there was no chunk taken out of either tool.
So, I re-shorted again, and this time left it on for 5 seconds. The CCA began to come up:22CCA.
I shorted for another 5 seconds, and got another gain: 28CCA.
With the tools getting warm, here is the last time:35.39CCA. It is nowhere near the original. Should be 500 or 600CCA.
So, I grabbed a couple of steel tools: a crescent and a socket bar. I shorted the posts together.
I was amazed to see just a tiny spark. I did not even need to blink, and there was no chunk taken out of either tool.
So, I re-shorted again, and this time left it on for 5 seconds. The CCA began to come up:22CCA.
I shorted for another 5 seconds, and got another gain: 28CCA.
With the tools getting warm, here is the last time:35.39CCA. It is nowhere near the original. Should be 500 or 600CCA.
I decided to bring the battery back up to full again, for further tests.
I applied 10 amps from the solar pannels for about 4 minutes.
And to my amazement, the voltage was already up to 13.3 volts. Way too short of a time!
Something is bad wrong! If I did not already know that...
I decided to look at Equalization:
I began to bring the voltage of the battery up, anticipating a target voltage of about 15 volts.
But before I could get there, the cells began to bubble at only 14.5 volts. That is too early!
Then I saw it! I saw the problem: Number 4 cell was not bubbling.
All the other cells were bubbling nicely, but number 4 was not bubbling at all.
All the cells were equalized, except number 4.
By the low gassing voltage of 14.4, I knew that a cell was shorted, and that cell was number 4.
I threw the battery in the car, and headed over to InterState Batteries. I told the guy that there was something wrong with number 4.
The guy sucked up electrolyte with his hydrometer. "Yes", he confirmed: number 4 is dead. Reads zero. Others great.
The Interstate guy explained that the battery was two years old, and the guarantee was only for one year.
It is a deepcycle. We guarantee Deepcycles for one year. And no Prorate beyond that year. SOL.
Obviously, time to go Lithium!
But...
There is a problem...
RV Battery B, #2, Right
 2019.11.29
2019.11.29
A new Interstate battery in Position #2.
This is Battery-Right, Battery #2.
I did not want to buy another Leadacid, but I had no choice. My Low Voltage System uses two Lead Acids.
The pair has to be the same chemistry and output voltage.
If I had a Lithium and a Leadacid together in the Low Voltage System, then the Lithium would be doing all the work.
It is not as bad as if the two batteries were connected together in parallel, which of course will not work, but almost as bad.
You see, in Automatic Mode, the Regulators will not know how to share the loading between the two different batteries.
And I like the idea of redundancy, so I am forced to buy it.
 Todays date is: 2019.11.29
Todays date is: 2019.11.29
The stamped date code is K90FP.
The "K" is for the 11th month.
The "9" is for the year 2019.
This is both a new battery this month, and fresh in my hands this month.
 Initial Charge looks good. 12.67 Volts
Initial Charge looks good. 12.67 Volts
4.20mOhms
 655 CCA
655 CCA
RV Battery A, #1, Left
Battery-Left, Battery #1
The factory delivered this battery 2015.June. Bought on 2015.09.20. Over 4 years old.
 5.60mOhms
5.60mOhms
Two years ago 2017 4.28mOhms, 691.0CCA
Ohms getting worse...
 507CCA
507CCA
Two years ago 2017 4.28mOhms, 691.0CCA
CCA getting worse. 73% is 58 AH now.
RV Battery A, #1
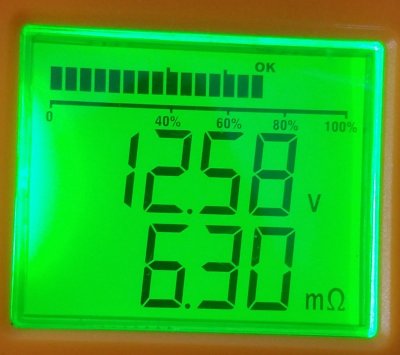
 2021.12.25
2021.12.25
Battery-A-#1, New 2015.09, 6 years old now
After a "cook" charge.
Battery has been revived by cooking: gassing all cells and equalizing all cells at top end, but still performance will not justify carrying around this much
extra weight.
RV Battery A, #1
I have now replaced this battery with a $320 100AH Lithium. (Hope it does not catch fire.)


 2021.12.27
2021.12.27
Battery-B-#2, New 2019.11, two years old now
Freshly Out of RV before analysis.

 2021.12.28
2021.12.28
Battery-B-#2
Battery cooked on the battery charger to see all cells gassing.
Original ohms was 4.20 mOhms, now 5.49 mOhms
I can now mix and match different chemistries. A mix of Lead and Lithium batteries is just fine.
So I will keep it for now.

 Put on the charger to gas 3 more hours...
Put on the charger to gas 3 more hours...
All cells equal in bubbles.
Brought the voltage up 13.23v on multimeter too. Good agreement.
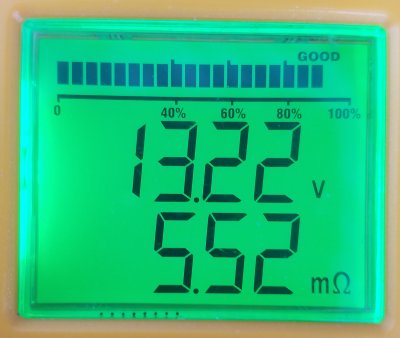
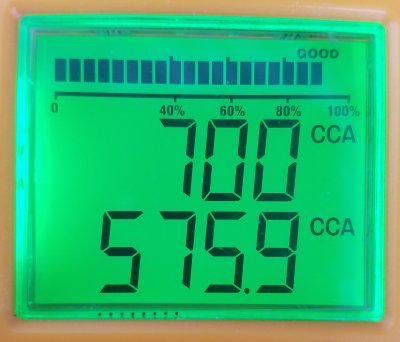 Changed the battery meter CCA setting to 700A just to see what would happen
Changed the battery meter CCA setting to 700A just to see what would happen
In just a few minutes voltage is starting to drop, as expected.
I have battery readings and histories that I can access in any campground or RV Park.
New Dodge Truck
Truck Battery:

 New Replacement Battery:
New Replacement Battery:
Todays date is 2023.05.12, the day of the replacement.
The old battery was exactly two years old, and was sold in a brand new 2021 truck.
The old battery would read 12.0 volts after sitting all night. But would usually start the truck.
The old battery:

 But the CCA seams perfect at 737 Amps.
But the CCA seams perfect at 737 Amps.
The dealership called on the phone and said the battery was "good", and not going to give me a new battery.
I said it would fail a voltage test in an hour or two of sitting.
The dealership kept the truck all night, and called in the morning, and said yes, it had failed.
I am glad the dealership has seen the problem, but being an engineer, I also know this failure mode is imposible without a parasitic draw.
There is no parasitic draw because the battery voltage-drop stops dropping at 12.0v.
And it is not a bad cell. 12.0V is a half cell. Imposible! Each cell is 2.1 volts. That would be 10.5 volts.
Therefor, there must be a relationship between the 48v system to the 12v system. Can one drain/charge the other?
 So, they gave me a new battery:
So, they gave me a new battery:
I am going to guess the year, as transparency is never ment for the consumer: The year is probably "2L", which is 2022.11.
I consider myself a consumer, and as such, am not privy to the decifer. This is a guess, at best.
If correct, it is fine; Only a few months old.
There is a 430 watt-hour lithium-ion nickel manganese cobalt (NMC)-graphite battery for the E-Torque motor-generator.
Van Battery
Van Battery:
 The old van battery is 11 years old.
The old van battery is 11 years old.
 128 CCA. Just clicks the starter.
128 CCA. Just clicks the starter.

 New Interstate for the Van.
New Interstate for the Van.


 Installing a regulator
Installing a regulator
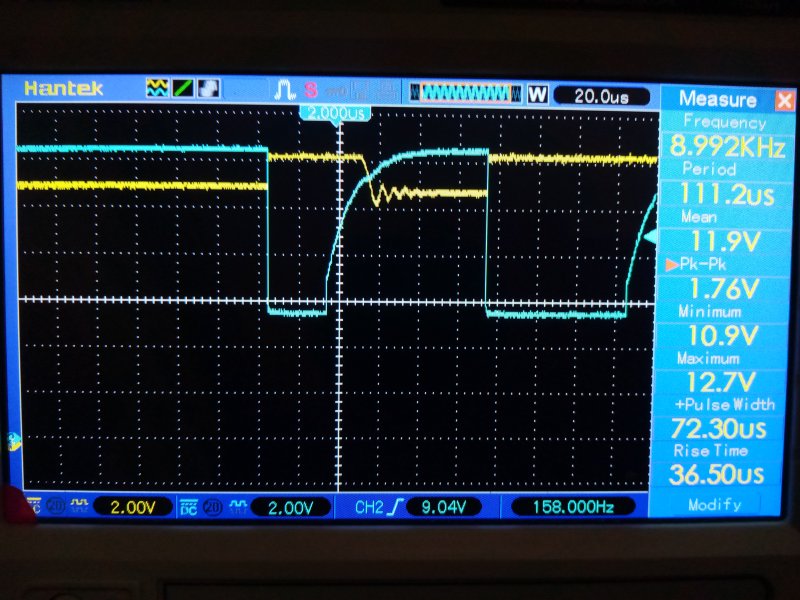 Waveform looks good.
Waveform looks good.
 Parasitic draw starts out very high at 0.2 Amps
Parasitic draw starts out very high at 0.2 Amps
But after 30 seconds, settles to 0.046 Amps, 46mA.
That is OK. I assume it is the security system.

Van Solar Panels
 Here is the back side of the 15 Watt Solar Panel.
Here is the back side of the 15 Watt Solar Panel.
There is heavy hail damage, that has penitrated to the bottom of the panel.
How this panel is still working, I do not know.
It produces over 0.7 Amps in ShortCircuit.
Originally, when new, it was 0.89A
0.7/0.9=77% Heavy degredation or hail damage.
 Removed the 15W Solar Panel from van.
Removed the 15W Solar Panel from van.
You can see the yellow patturn. It is permanent. It is burned-in.
Silicone had been used to secure the panel.
 Unfortunately, Silicone had been used to secure the panel.
Unfortunately, Silicone had been used to secure the panel.
Today, I never use silicone. It can not withstand high heat, as is found on a van.
The panel came off very easily, with only a gentle pull with the hands.
I was expecting to use a scraper, as young silicone is almost imposible to remove.
Here the silicone has degraded to fragile pieces, that can be gently scraped off with a fingernail, or in some cases brushed away.
Silicone is terrible. My panel could have flown off.
 I removed this 30W panel from the RV.
I removed this 30W panel from the RV.
Here is the 30W Solar Panel ShortCircuit with 1.62A
This panel was new in 2017.10.12 from Ebay
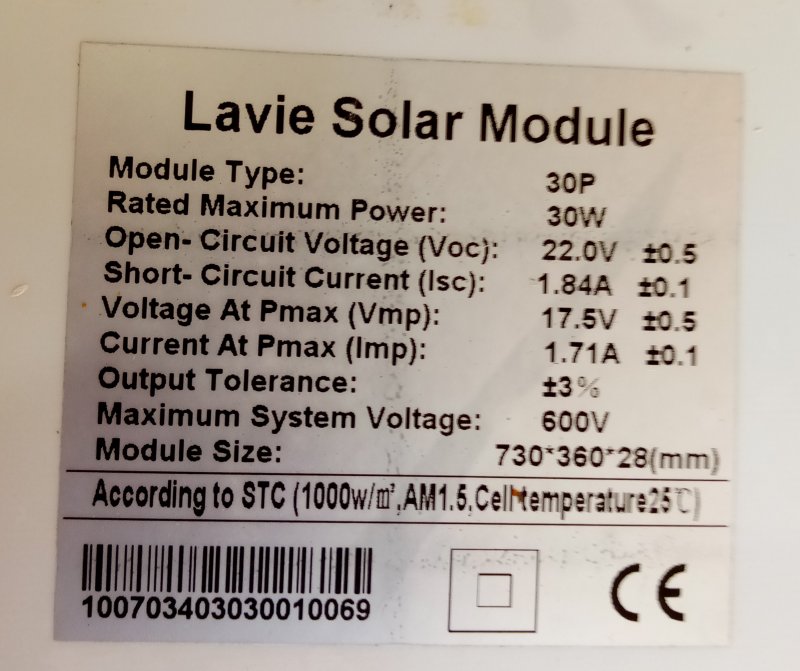 Here is the specks on the 30W Panel.
Here is the specks on the 30W Panel.
Original ShortCircuit of 1.84Amps in 2017.10.12.
1.62/1.84 = 88% in 6 years.




 Voltage at the posts under an initial 5.0 amp load.
Voltage at the posts under an initial 5.0 amp load.
 This battery failed in less than 5 months. Capacity continually dropped until now only about 10AHr.
This battery failed in less than 5 months. Capacity continually dropped until now only about 10AHr. Purchased this battery locally, at Ron's RV.
Purchased this battery locally, at Ron's RV. The date code shows that the battery is mine: My initials are "CAP", Cecil Alan Pennock.
Just kidding; Wierd things happen all the time.
For example, before custom car plates in the 50's,
my Dad had a licence plate - quite by accident - that was "Cec". Coincidently, he went by that name!
But I do not go by "CAP".
The date code shows that the battery is mine: My initials are "CAP", Cecil Alan Pennock.
Just kidding; Wierd things happen all the time.
For example, before custom car plates in the 50's,
my Dad had a licence plate - quite by accident - that was "Cec". Coincidently, he went by that name!
But I do not go by "CAP".
 84 AHr
84 AHr





 Got the bucket of water out. Found two one ohm resistors.
Wired them in series, and added a little incandescent bulb in parallel, for esthetics.
The glow on the left is the incandescent under the water.
Got the bucket of water out. Found two one ohm resistors.
Wired them in series, and added a little incandescent bulb in parallel, for esthetics.
The glow on the left is the incandescent under the water.
 Placed everything in the bucket of water. Everything reads about 1.5 ohms.
Aiming for about 6 amps.
The standard 20C would be in this case 4 amps, but I want to hurry this up.
Placed everything in the bucket of water. Everything reads about 1.5 ohms.
Aiming for about 6 amps.
The standard 20C would be in this case 4 amps, but I want to hurry this up.

 On 2017.09.25
On 2017.09.25 Batt #2
Batt #2 Standard Group 24
Standard Group 24
 State of Charge: 12.66volts
State of Charge: 12.66volts
 Here is another set of readings, after setting half hour.
Here is another set of readings, after setting half hour.


 After charging for a day at 13.40 volts, the battery was disconnected.
And almost immediatly it dropped to 11.23 volts. It has to have a bad cell...
And, it has only 16 CCA instead of 600CCA. Something is bad wrong!
After charging for a day at 13.40 volts, the battery was disconnected.
And almost immediatly it dropped to 11.23 volts. It has to have a bad cell...
And, it has only 16 CCA instead of 600CCA. Something is bad wrong!
 Rechecked the meter. It is even worse: 11CCA.
Rechecked the meter. It is even worse: 11CCA. So, I grabbed a couple of steel tools: a crescent and a socket bar. I shorted the posts together.
I was amazed to see just a tiny spark. I did not even need to blink, and there was no chunk taken out of either tool.
So, I re-shorted again, and this time left it on for 5 seconds. The CCA began to come up:22CCA.
I shorted for another 5 seconds, and got another gain: 28CCA.
With the tools getting warm, here is the last time:35.39CCA. It is nowhere near the original. Should be 500 or 600CCA.
So, I grabbed a couple of steel tools: a crescent and a socket bar. I shorted the posts together.
I was amazed to see just a tiny spark. I did not even need to blink, and there was no chunk taken out of either tool.
So, I re-shorted again, and this time left it on for 5 seconds. The CCA began to come up:22CCA.
I shorted for another 5 seconds, and got another gain: 28CCA.
With the tools getting warm, here is the last time:35.39CCA. It is nowhere near the original. Should be 500 or 600CCA.
 2019.11.29
2019.11.29 Todays date is: 2019.11.29
Todays date is: 2019.11.29 Initial Charge looks good. 12.67 Volts
Initial Charge looks good. 12.67 Volts 655 CCA
655 CCA 5.60mOhms
5.60mOhms  507CCA
507CCA 
 2021.12.25
2021.12.25

 2021.12.27
2021.12.27
 2021.12.28
2021.12.28
 Put on the charger to gas 3 more hours...
Put on the charger to gas 3 more hours...
 Changed the battery meter CCA setting to 700A just to see what would happen
Changed the battery meter CCA setting to 700A just to see what would happen
 New Replacement Battery:
New Replacement Battery:
 But the CCA seams perfect at 737 Amps.
But the CCA seams perfect at 737 Amps.  So, they gave me a new battery:
So, they gave me a new battery: The old van battery is 11 years old.
The old van battery is 11 years old.
 128 CCA. Just clicks the starter.
128 CCA. Just clicks the starter.

 New Interstate for the Van.
New Interstate for the Van.


 Installing a regulator
Installing a regulator
 Waveform looks good.
Waveform looks good.
 Parasitic draw starts out very high at 0.2 Amps
Parasitic draw starts out very high at 0.2 Amps  Here is the back side of the 15 Watt Solar Panel.
Here is the back side of the 15 Watt Solar Panel.  Removed the 15W Solar Panel from van.
Removed the 15W Solar Panel from van.  Unfortunately, Silicone had been used to secure the panel.
Unfortunately, Silicone had been used to secure the panel.  I removed this 30W panel from the RV.
I removed this 30W panel from the RV. Here is the specks on the 30W Panel.
Here is the specks on the 30W Panel.